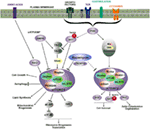
This review describes the components of protein scaffolds, different ways of constructing a protein scaffold-based multi-enzyme complex, and their effects on enzyme kinetics. The scaffolding improves the catalytic performance, enzyme stability and provides an optimal micro-environment for biochemical reactions. With this respect, scaffolding proteins play an immense role in bringing different enzymes together in a specific manner. However, operating different enzymes together in a single vessel limits their operational performance which needs to be addressed. It seems likely that further scaffold proteins will be identified in other protein kinase signaling pathways in the future.The synthesis of complex molecules using multiple enzymes simultaneously in one reaction vessel has rapidly emerged as a new frontier in the field of bioprocess technology. The principle of scaffolding kinases is not restricted to MAP kinase pathways, but includes the scaffold protein IKAP that organizes three protein kinases (NIK, IKKa, and IKKP) required for activation of the NFkB transcription factor as well as AKAPS for the cAMP-dependent protein kinase. The conservation of scaffolds in the pathways mentioned here is not too surprising as all kinases in question are related members of the MAP kinase family. Another protein, MPl, seems to play a similar role by bundling two other protein kinases, MEKl and ERKl. In mammals, JIPl may play a similar role to Ste5p in linking three protein kinases-MLK3, MKK7, and JNK-that are sequentially activated by cytokines or stress. Conversely, in the osmolarity response pathway, the protein kinase Pbs2p seems to double up as a scaffold by linking its activator Stellp with its substrate Hoglp. This was due to its association with the scaffold protein Ste5p in the mating response that aligns Stellp with its target Ste7p, another kinase, and Fus3p, a third protein kinase that is regulated by Ste7p. However, Stellp activated by mating pheromones will not activate the osmolarity response, and vice versa. Tein kinase that regulates the activity of other kinases further down the line, in both the mating response and osmolarity response pathways. Assembly of protein kinases into complexes through scaffold proteins prevents Stellp activation by pheromone to cross-activate the osmolarity response, and conversely, Stellp activation by high osmolarity will not activate the mating response. In this pathway, Pbs2p serves a dual function as kinase and scaffold protein. Stellp is also part of a protein kinase cascade activated in response to high extracellular osmolarity, together with Pbs2p and Hoglp kinases. They are linked into a complex by the Ste5p scaffold protein. In yeast, Stellp, Ste7p, and Fus3p form a protein kinase cascade leading to serial activation in the response to pheromones. Regulation of protein kinase cascades by scaffold proteins. This was first noted in the yeast Saccharomyces cerevisiae, where Ste11p acts as an "upstream" pro-įig. It has become apparent that several protein kinases are organized into functional signalling complexes by stable association with so-called scaffold proteins (see Fig. Whether protein kinases phosphorylate their substrates or not is not only determined by their activation, but also by their access to their targets.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
